Chlorine And Bromine Ionic Or Covalent . note, members of the same family tend to form similar compounds, so bromine and iodine form similar anions to chlorine (see. aluminum and chlorine react to form an ionic compound. for example, consider binary ionic compounds of iron and chlorine. Iron typically exhibits a charge of either 2+ or. Iron typically exhibits a charge of either 2+ or 3+ (see figure 2 in chapter 4.2 ionic and. for example, consider binary ionic compounds of iron and chlorine. Predict which forms an anion, which forms a cation, and the charges of. covalent bonds are the attractive forces between the positively charged nuclei of the bonded atoms and one or more pairs of. when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet.
from stock.adobe.com
Iron typically exhibits a charge of either 2+ or 3+ (see figure 2 in chapter 4.2 ionic and. for example, consider binary ionic compounds of iron and chlorine. aluminum and chlorine react to form an ionic compound. covalent bonds are the attractive forces between the positively charged nuclei of the bonded atoms and one or more pairs of. Predict which forms an anion, which forms a cation, and the charges of. when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. Iron typically exhibits a charge of either 2+ or. note, members of the same family tend to form similar compounds, so bromine and iodine form similar anions to chlorine (see. for example, consider binary ionic compounds of iron and chlorine.
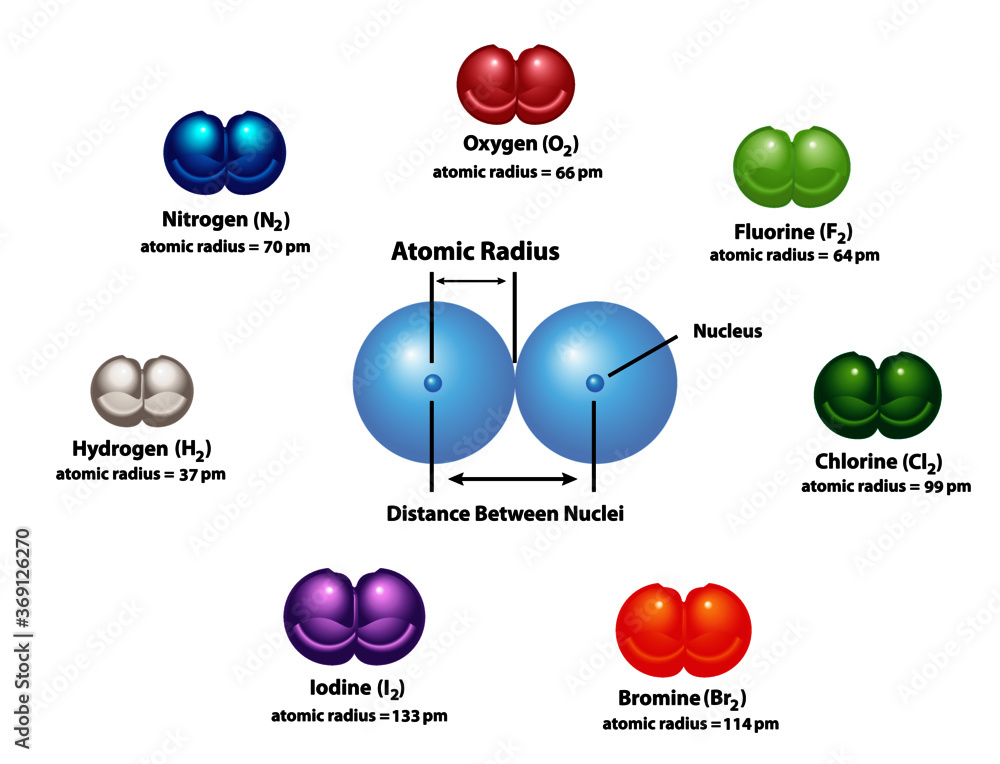
Diagram explaining Atomic Radius using diatomic molecules. Oxygen, hydrogen, nitrogen, fluorine
Chlorine And Bromine Ionic Or Covalent note, members of the same family tend to form similar compounds, so bromine and iodine form similar anions to chlorine (see. Predict which forms an anion, which forms a cation, and the charges of. for example, consider binary ionic compounds of iron and chlorine. note, members of the same family tend to form similar compounds, so bromine and iodine form similar anions to chlorine (see. Iron typically exhibits a charge of either 2+ or. when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. covalent bonds are the attractive forces between the positively charged nuclei of the bonded atoms and one or more pairs of. aluminum and chlorine react to form an ionic compound. Iron typically exhibits a charge of either 2+ or 3+ (see figure 2 in chapter 4.2 ionic and. for example, consider binary ionic compounds of iron and chlorine.
From www.buzzle.com
Bromine Vs. Chlorine Chlorine And Bromine Ionic Or Covalent when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. Iron typically exhibits a charge of either 2+ or. note, members of the same family tend to form similar compounds, so bromine and iodine form similar anions to chlorine (see. Iron typically exhibits a charge of either 2+ or. Chlorine And Bromine Ionic Or Covalent.
From www.diffzy.com
Bromine vs. Chlorine What's The Difference In Tabular Form, Points, Definitions, Examples Chlorine And Bromine Ionic Or Covalent for example, consider binary ionic compounds of iron and chlorine. Predict which forms an anion, which forms a cation, and the charges of. when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. aluminum and chlorine react to form an ionic compound. covalent bonds are the attractive. Chlorine And Bromine Ionic Or Covalent.
From exofhdgch.blob.core.windows.net
Bromine Ionic Or Covalent at Donna McMillian blog Chlorine And Bromine Ionic Or Covalent covalent bonds are the attractive forces between the positively charged nuclei of the bonded atoms and one or more pairs of. note, members of the same family tend to form similar compounds, so bromine and iodine form similar anions to chlorine (see. Iron typically exhibits a charge of either 2+ or. when forming ions, elements typically gain. Chlorine And Bromine Ionic Or Covalent.
From diagramdataperianths.z21.web.core.windows.net
Diagram Ionic Bond Chlorine And Bromine Ionic Or Covalent for example, consider binary ionic compounds of iron and chlorine. covalent bonds are the attractive forces between the positively charged nuclei of the bonded atoms and one or more pairs of. Predict which forms an anion, which forms a cation, and the charges of. aluminum and chlorine react to form an ionic compound. Iron typically exhibits a. Chlorine And Bromine Ionic Or Covalent.
From revisechemistry.uk
Chemical Bonds, Ionic, Covalent and Metallic AQA C2 revisechemistry.uk Chlorine And Bromine Ionic Or Covalent for example, consider binary ionic compounds of iron and chlorine. Iron typically exhibits a charge of either 2+ or 3+ (see figure 2 in chapter 4.2 ionic and. Iron typically exhibits a charge of either 2+ or. aluminum and chlorine react to form an ionic compound. note, members of the same family tend to form similar compounds,. Chlorine And Bromine Ionic Or Covalent.
From askanydifference.com
Bromine vs Chlorine Difference and Comparison Chlorine And Bromine Ionic Or Covalent Iron typically exhibits a charge of either 2+ or. when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. Iron typically exhibits a charge of either 2+ or 3+ (see figure 2 in chapter 4.2 ionic and. for example, consider binary ionic compounds of iron and chlorine. Predict which. Chlorine And Bromine Ionic Or Covalent.
From pediaa.com
Difference Between Bromine and Chlorine Chlorine And Bromine Ionic Or Covalent for example, consider binary ionic compounds of iron and chlorine. Iron typically exhibits a charge of either 2+ or. note, members of the same family tend to form similar compounds, so bromine and iodine form similar anions to chlorine (see. for example, consider binary ionic compounds of iron and chlorine. aluminum and chlorine react to form. Chlorine And Bromine Ionic Or Covalent.
From www.revisechemistry.uk
Bonding and Properties of materials OCR Gateway C2 revisechemistry.uk Chlorine And Bromine Ionic Or Covalent covalent bonds are the attractive forces between the positively charged nuclei of the bonded atoms and one or more pairs of. Iron typically exhibits a charge of either 2+ or 3+ (see figure 2 in chapter 4.2 ionic and. Iron typically exhibits a charge of either 2+ or. Predict which forms an anion, which forms a cation, and the. Chlorine And Bromine Ionic Or Covalent.
From slideplayer.com
The Chemistry of Life Chapter 2 ppt download Chlorine And Bromine Ionic Or Covalent Predict which forms an anion, which forms a cation, and the charges of. aluminum and chlorine react to form an ionic compound. for example, consider binary ionic compounds of iron and chlorine. note, members of the same family tend to form similar compounds, so bromine and iodine form similar anions to chlorine (see. when forming ions,. Chlorine And Bromine Ionic Or Covalent.
From www.numerade.com
SOLVED Which of the pairs of atoms below would be expected to make covalent bonds? Select all Chlorine And Bromine Ionic Or Covalent aluminum and chlorine react to form an ionic compound. covalent bonds are the attractive forces between the positively charged nuclei of the bonded atoms and one or more pairs of. note, members of the same family tend to form similar compounds, so bromine and iodine form similar anions to chlorine (see. for example, consider binary ionic. Chlorine And Bromine Ionic Or Covalent.
From stock.adobe.com
Vecteur Stock Diatomic molecules diagram shows elements that exist as diatomic molecules. Oxygen Chlorine And Bromine Ionic Or Covalent Iron typically exhibits a charge of either 2+ or. aluminum and chlorine react to form an ionic compound. for example, consider binary ionic compounds of iron and chlorine. covalent bonds are the attractive forces between the positively charged nuclei of the bonded atoms and one or more pairs of. Predict which forms an anion, which forms a. Chlorine And Bromine Ionic Or Covalent.
From www.numerade.com
SOLVED Using the electronegativity chart, identify the bond type between chlorine (Cl) and Chlorine And Bromine Ionic Or Covalent note, members of the same family tend to form similar compounds, so bromine and iodine form similar anions to chlorine (see. covalent bonds are the attractive forces between the positively charged nuclei of the bonded atoms and one or more pairs of. Predict which forms an anion, which forms a cation, and the charges of. aluminum and. Chlorine And Bromine Ionic Or Covalent.
From chemistrylearnwithsangam.blogspot.com
chemistry knowledge Comparison between Covalent and Ionic Bond Chlorine And Bromine Ionic Or Covalent Iron typically exhibits a charge of either 2+ or 3+ (see figure 2 in chapter 4.2 ionic and. aluminum and chlorine react to form an ionic compound. Iron typically exhibits a charge of either 2+ or. note, members of the same family tend to form similar compounds, so bromine and iodine form similar anions to chlorine (see. . Chlorine And Bromine Ionic Or Covalent.
From www.poolsuppliessuperstore.com
Bromine vs. Chlorine Everything You Need to Know Pool Supplies Superstore Chlorine And Bromine Ionic Or Covalent for example, consider binary ionic compounds of iron and chlorine. for example, consider binary ionic compounds of iron and chlorine. aluminum and chlorine react to form an ionic compound. Predict which forms an anion, which forms a cation, and the charges of. covalent bonds are the attractive forces between the positively charged nuclei of the bonded. Chlorine And Bromine Ionic Or Covalent.
From www.alamy.com
Fluorine, Bromine and Chlorine Molecular Model of Atom. Vector illustration Stock Vector Image Chlorine And Bromine Ionic Or Covalent when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. note, members of the same family tend to form similar compounds, so bromine and iodine form similar anions to chlorine (see. Predict which forms an anion, which forms a cation, and the charges of. Iron typically exhibits a charge. Chlorine And Bromine Ionic Or Covalent.
From questions-in.kunduz.com
4.34 Identify the bonds formed between t... Chemistry Chlorine And Bromine Ionic Or Covalent note, members of the same family tend to form similar compounds, so bromine and iodine form similar anions to chlorine (see. Iron typically exhibits a charge of either 2+ or 3+ (see figure 2 in chapter 4.2 ionic and. for example, consider binary ionic compounds of iron and chlorine. when forming ions, elements typically gain or lose. Chlorine And Bromine Ionic Or Covalent.
From stock.adobe.com
Diagram explaining Atomic Radius using diatomic molecules. Oxygen, hydrogen, nitrogen, fluorine Chlorine And Bromine Ionic Or Covalent Predict which forms an anion, which forms a cation, and the charges of. covalent bonds are the attractive forces between the positively charged nuclei of the bonded atoms and one or more pairs of. Iron typically exhibits a charge of either 2+ or 3+ (see figure 2 in chapter 4.2 ionic and. for example, consider binary ionic compounds. Chlorine And Bromine Ionic Or Covalent.
From resource.studiaacademy.com
Group 7 (Halogens) Chlorine, Bromine and Iodine Studia Academy Resources Chlorine And Bromine Ionic Or Covalent Iron typically exhibits a charge of either 2+ or 3+ (see figure 2 in chapter 4.2 ionic and. Iron typically exhibits a charge of either 2+ or. Predict which forms an anion, which forms a cation, and the charges of. covalent bonds are the attractive forces between the positively charged nuclei of the bonded atoms and one or more. Chlorine And Bromine Ionic Or Covalent.